Understanding the Condition
What is an Osteochondral Lesion of the Talus (OLT)?
Osteochondral lesions of the talus involve damage to the articular cartilage and underlying bone of the talus (ankle bone). These lesions pose a therapeutic challenge due to the limited intrinsic healing capacity of cartilage and the talus's unique anatomical characteristics [1]:
- The talus has a large articular surface area (60% of total talar body surface) [2]
→ Articular surface = the smooth, slippery coating where bones meet in a joint
- It is devoid of soft tissue attachment and lacks sufficient blood supply [3]
→ No muscles/tendons attach directly to the talus, and blood flow is limited - both hurt healing
- This leads to poor healing and complications such as osteonecrosis [4]
→ Osteonecrosis = bone death from lack of blood supply
The ankle is the second most frequent site, following the knee, that requires cartilage repair [5].
Size Classification: Why It Matters
OLTs are generally classified as small or large based on:
- Area: Greater or less than 1.5 cm² (150 mm²) [6]
- Diameter: Greater or less than 15 mm [7]
A study in the American Journal of Sports Medicine identified 150 mm² as a critical cutoff for clinical failure. Only 10.5% of ankles with defects smaller than 150 mm² showed clinical failure, compared to significantly higher rates in larger lesions [6].
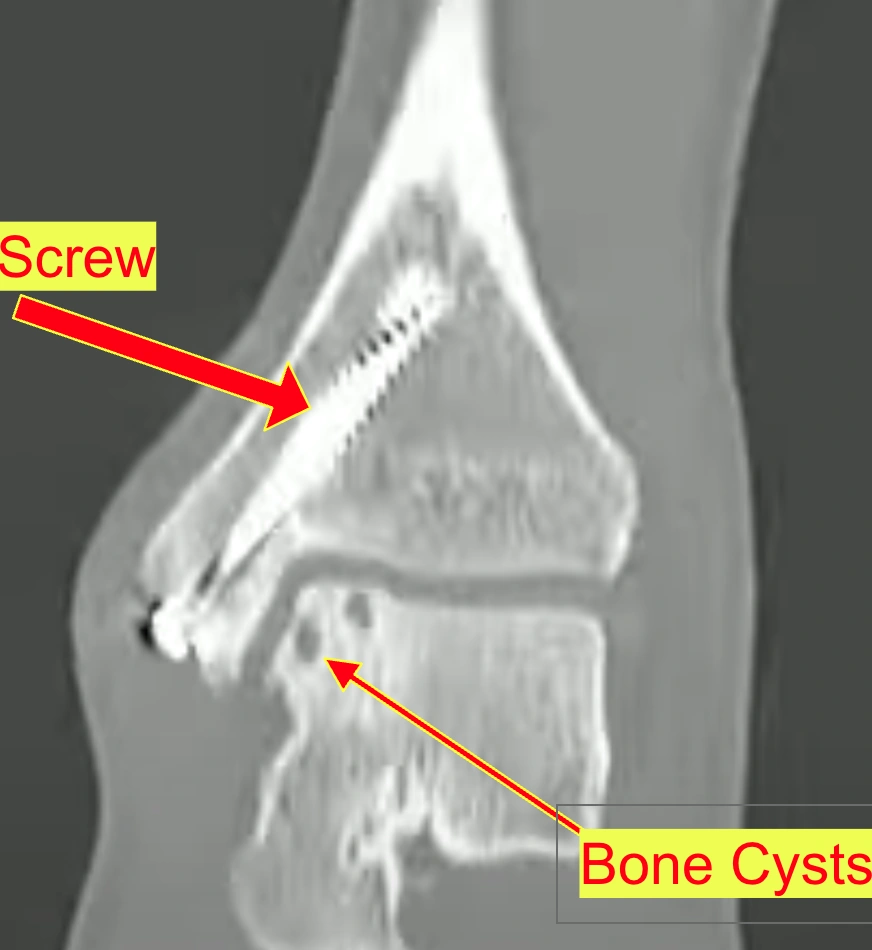
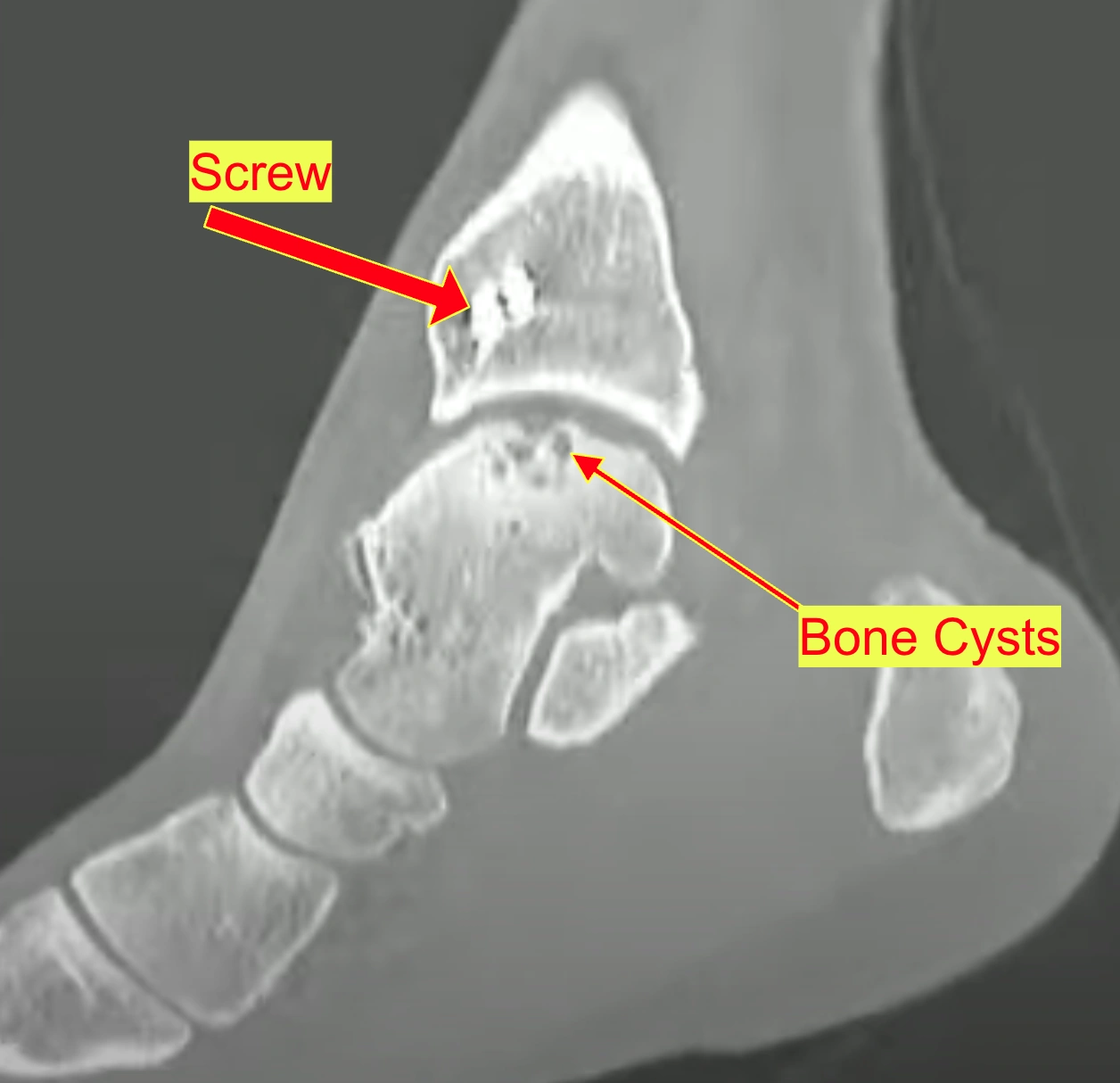
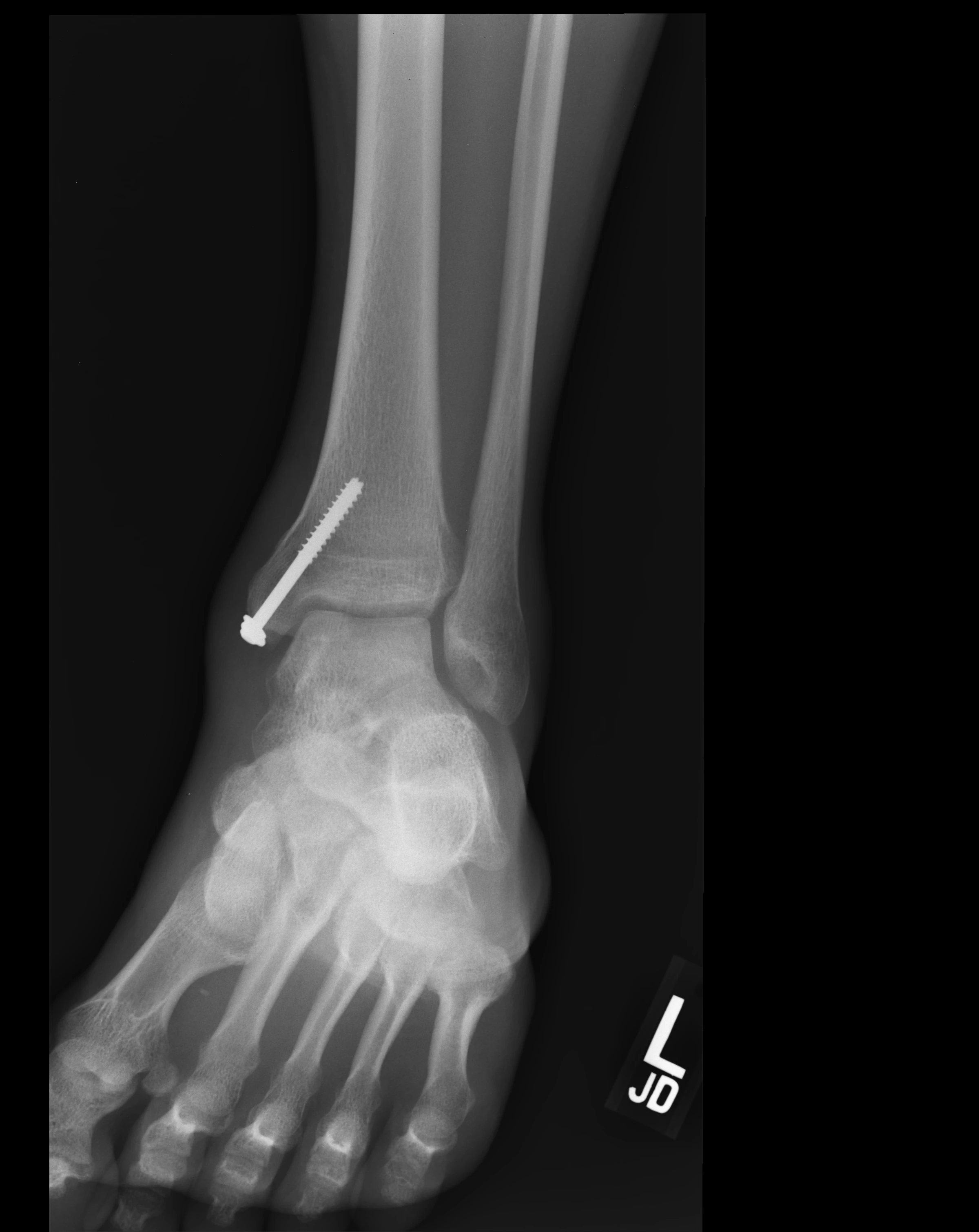
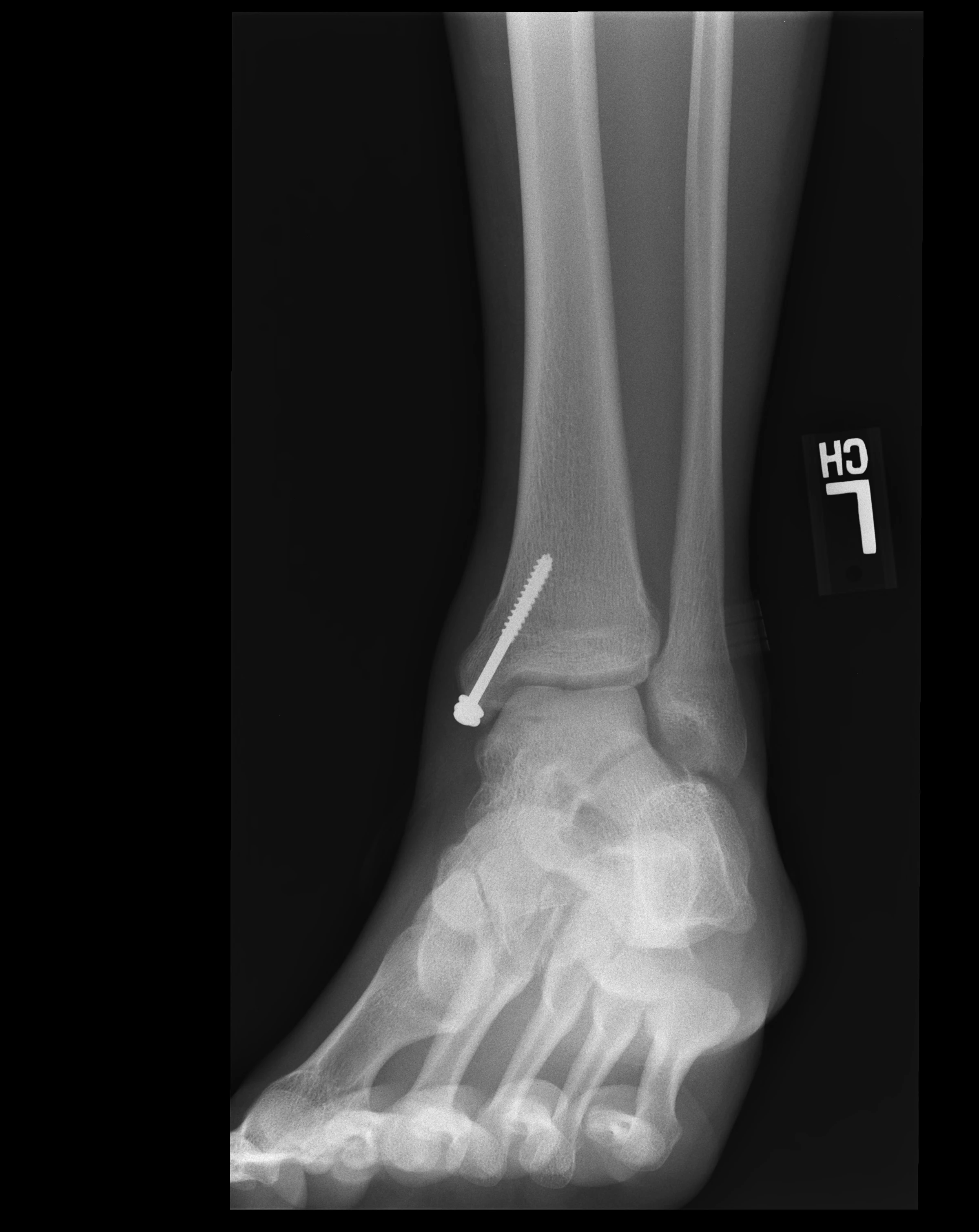
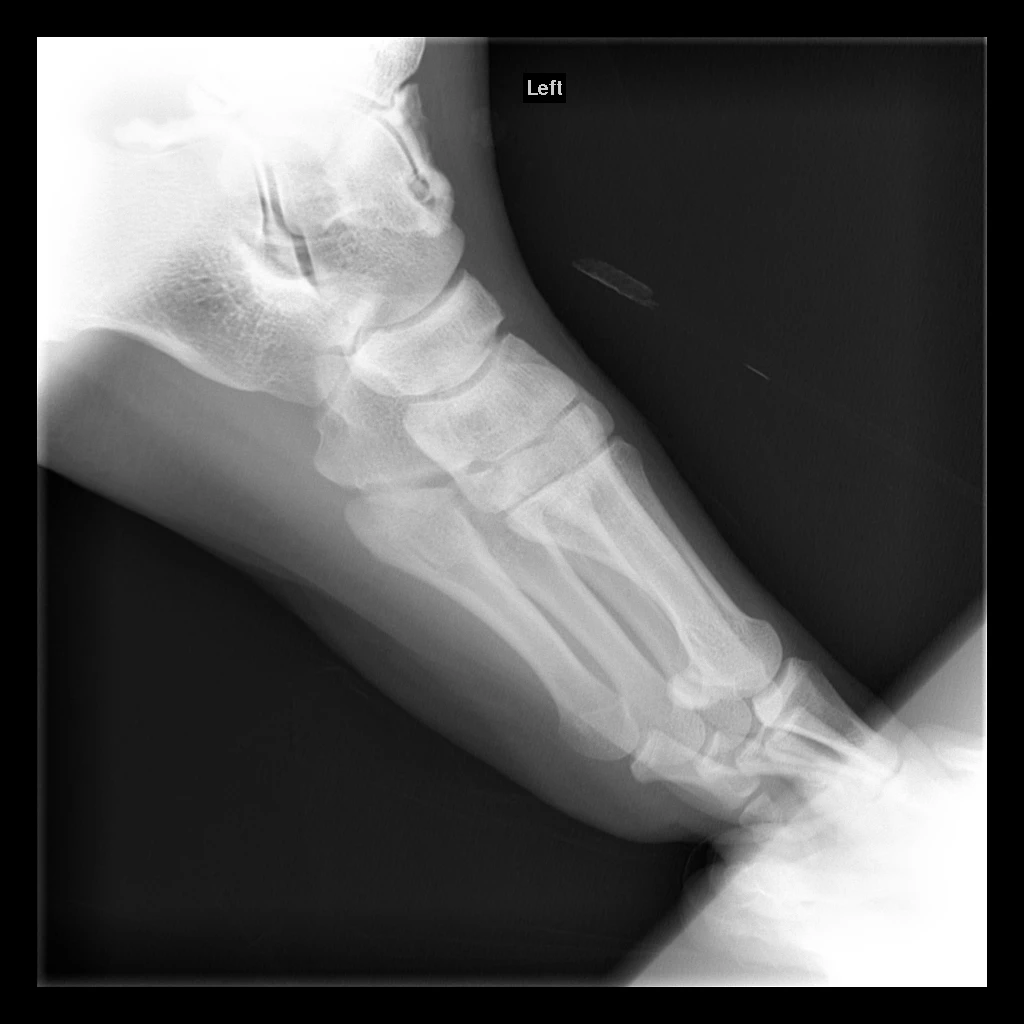
Raymond's lesion measures approximately 15mm x 10mm for cartilage loss and 15mm x 12mm x 10mm for the cystic bone component, placing it in the "large lesion" category.
Natural History of Untreated OLT
Multiple studies have examined what happens when OLTs are not surgically treated:
- A 14-year follow-up study found 88% of patients who successfully completed non-operative treatment remained minimally symptomatic (VAS 0-3) [8]
→ VAS = Visual Analog Scale for pain (0 = no pain, 10 = worst pain imaginable)
- Only 6% needed surgical intervention in the long term if initial conservative treatment was successful [8]
- Progression of ankle osteoarthritis by 1 grade was seen in 27% of cases, while 73% showed no deterioration [8]
→ Osteoarthritis = "wear and tear" arthritis where cartilage breaks down over time
- However, a decrease in sports activity due to ankle pain was observed in more than one-third of patients [9]
MRI Staging Classifications
→ MRI staging = how doctors classify OLT severity. Higher stages = more damage = likely need surgery. Lower stages may heal conservatively.
Several classification systems exist for staging OLT on imaging:
Berndt and Harty Classification (1959) - Most Commonly Used
- Stage 1: Subchondral bone compression (no visible lesion on plain X-ray)
→ Subchondral = the bone layer just beneath the cartilage surface
- Stage 2: Partially detached osteochondral fragment
→ Fragment = a piece of cartilage+bone starting to separate but still attached
- Stage 3: Completely detached but undisplaced fragment
→ Piece is loose but sitting in its original spot
- Stage 4: Completely detached and displaced fragment
→ Loose piece has moved from its original location (like a loose pebble in a shoe)
- Stage 5 (Loomer modification): Subchondral cyst present [120]
→ A fluid-filled hole has formed in the bone beneath the cartilage
Hepple Classification (1999) - MRI-Specific
Hepple and colleagues revised the classification specifically for MRI findings, noting that 30-43% of OLTs visible on MRI are invisible on conventional radiography [121]. MRI can detect early biochemical changes before morphological damage appears.
→ Radiography = standard X-rays; morphological = visible structural changes
Quantitative MRI: T2 Mapping (2024-2025)
Advanced imaging techniques now allow quantitative assessment of cartilage quality [122]:
- T2 mapping quantifies water and collagen changes within cartilage
→ T2 mapping = MRI technique measuring water content in cartilage; collagen = protein fibers giving cartilage strength
- Enables detection of early damage before visible morphological changes
- Lower T2 values after treatment may indicate more hyaline-like (better quality) repair tissue
→ Hyaline cartilage = the original "glass-smooth" cartilage type; what we want to restore
Raymond's lesion has been classified as Hepple Stage V (with cystic component), placing it in the most severe category.
Normal Talar Cartilage Thickness
→ Understanding normal = understanding how much damage exists. Talar cartilage is thin (about 1mm) - even small defects are significant.
MRI and cadaveric studies have established normal talar cartilage thickness values [123]:
→ Cadaveric = studied from deceased donors; gives true measurements without imaging distortion
- Average thickness: 0.89-1.35 mm depending on location and measurement method
- Male specimens: 1.35 ± 0.22 mm average
- Female specimens: 1.11 ± 0.28 mm average
- Thickest area: Medial corner of talar dome
→ Medial = inner side (toward big toe); talar dome = rounded top of the talus bone
- Thinnest area: Lateral gutter
→ Lateral = outer side (toward pinky toe); gutter = groove along the edge
- MRI tends to overestimate thickness by 0.16-0.32 mm compared to direct measurement
Clinical significance: Smaller joints like the ankle have thinner cartilage, making MRI evaluation less accurate than in the knee.
Risk Factors for Treatment Failure (2024-2025 Evidence)
→ Certain things make treatments less likely to work. Knowing these helps set realistic expectations and optimize conditions before surgery.
Smoking
A 2025 study with minimum 5-year follow-up found [124]:
- Smokers have significantly worse pain and functional outcomes after osteochondral transplantation
- While both groups improve, non-smokers demonstrate superior clinical recovery
- Smoking cessation should be integrated into perioperative management
→ Perioperative = the period before, during, and after surgery
Obesity (BMI ≥30)
The 2024 JBJS 10-year survival study found [22]:
→ BMI = Body Mass Index, a weight-to-height ratio; ≥30 is considered obese
- BMI ≥30 significantly associated with higher likelihood of revision surgery
- Hazard ratio: 3.0 (95% CI: 1.44-6.43, p<0.01)
→ Hazard ratio 3.0 = obese patients are 3x more likely to need another surgery; CI = confidence interval; p<0.01 = statistically significant (not random chance)
- Central lesions more affected by increased BMI due to biomechanical overloading
→ Central lesions = damage in the middle of the joint surface where weight bearing is highest
Age
- Older age at consultation correlates with lower sports activity levels (Spearman = -0.52, p=0.01)
→ Spearman = statistical correlation test; negative value means older = less active
- Younger patients and smaller lesions are independent predictors of surgical success
- Patients ≥60 years old more commonly have concomitant varus deformity [125]
→ Concomitant = occurring together; varus deformity = ankle tilted inward (bowlegged alignment)
Lesion Size
- Critical cutoff: 150 mm² area for clinical failure risk [6]
- Lesions >125 mm² have significantly increased failure risk with some techniques [46]
Ankle Instability and OLT Relationship (2024-2025)
→ Wobbly ankle = damaged cartilage. Up to 70% of ankle sprains/fractures may cause OLT. Fixing instability protects cartilage repairs from failing.
Prevalence
Recent 2024-2025 studies reveal strong associations [126]:
- 31.78% of patients with anterior talofibular ligament (ATFL) injuries have concurrent OLT
→ ATFL = the ligament on the outside front of the ankle, most commonly sprained
- Up to 70% of ankle sprains and fractures may result in osteochondral lesions
- Chronic lateral ankle instability (CLAI) often leads to OLT and subsequent osteoarthritis
→ CLAI = ongoing ankle "giving way" or wobbliness from old sprains that didn't fully heal
Clinical Significance
Joint line tenderness lasting >6 weeks strongly predicts presence of OLT (p<0.001) [126]. Physical examination has low sensitivity for detecting OLT - MRI recommended for persistent symptoms.
→ Joint line tenderness = pain when pressing directly on the ankle joint; sensitivity = how well a test catches problems
Treatment Implications
A 2024-2025 study found [127]:
- Presence of CLAI negatively affects postoperative functional outcomes in OLT patients
- However, lateral ligament repair does not negatively impact cartilage repair outcomes
- Treating instability simultaneously is paramount for protecting repaired cartilage
- Preventing recurrent micro-instability minimizes shear forces and further degeneration
→ Shear forces = sliding/grinding stress on cartilage from an unstable joint
2025 Update: Alignment Assessment in OLT Planning
A 2025 study found that whole-leg mechanical axis alignment and talar tilt should be part of standard OLT surgical planning [157]:
- Concomitant CLAI lowered postoperative function even when cartilage repair itself was not clearly harmed once stability was restored
- Whole-leg alignment assessment (not just ankle-level) is now recommended before revision surgery
- In a revision ankle with a sprain history, this is not a side issue — it's a prerequisite
Clinical pearl: If you had significant ankle sprains before your OLT, addressing any residual instability is critical for long-term success of cartilage treatment. For revision cases especially, alignment must be checked before choosing a procedure.
Treatment Options: What the Evidence Says
1. Conservative Management
Success Rates (Aggregated from Multiple Systematic Reviews)
→ "Conservative" means managing without surgery: rest, PT, bracing, anti-inflammatories. The cartilage won't heal, but symptoms may calm down.
62%
Eventually convert to surgery
[10]
11%
Develop OA on imaging
[11]
88%
Pain-free at 14 yrs (if initially successful)
[8]
2025 Prospective Study (First of Its Kind)
The first prospective study assessing nonoperative treatment for OLTs was published in 2025 [13]:
→ Prospective study = follows patients forward in time (stronger evidence than looking backward)
- Nonoperative treatment yields statistically significant pain improvement after 1 year
- However, only 38% achieved clinically relevant improvement (exceeded MCID)
→ MCID = Minimal Clinically Important Difference; the smallest change patients actually notice
- Lesion size remained stable at 12-month follow-up
- Majority of improvements did not exceed minimal clinically important difference
2026 Long-Term Nonoperative Survival (CARTILAGE)
A 2026 study with median 66-month follow-up found [171]:
- 77% of OLT patients were successfully managed nonoperatively at 5+ years
- Survival rates: 93% at 1 year, 90% at 2 years, 77% at 5 years
- Each year older at diagnosis = 7% lower likelihood of converting to surgery
→ Older patients are less likely to eventually need surgery. For younger active patients, the odds of eventually needing surgery are higher.
What Conservative Treatment Includes
- Rest, restriction of activities, and immobilization (cast for 4-6 weeks if acute) [12]
- NSAIDs for pain and inflammation [1]
→ NSAIDs = Non-Steroidal Anti-Inflammatory Drugs (ibuprofen, naproxen, etc.)
- Physical therapy: peroneal strengthening, ROM, proprioceptive training [3]
→ Peroneal = muscles on outer lower leg; ROM = Range of Motion; proprioceptive = balance/position sense training
- Bracing/orthotics to decrease stress on the joint [63]
Return to Sports After Conservative Treatment
A 2024 systematic review of 2,347 cases found [64]:
- 76% continue participating in sports at long-term follow-up after arthroscopic treatment
→ Arthroscopic = using a tiny camera through small incisions (minimally invasive surgery)
- Activity level decreased and never reached pre-injury level
- 72.8% able to resume sports at preinjury level after surgical treatment
Juvenile/Pediatric OLT (2024-2025 Updates)
A 2025 systematic review on juvenile OLTs found [65]:
- Skeletally immature patients have high potential for spontaneous recovery
→ Skeletally immature = growth plates still open; kids' bones are still growing
- Conservative success rates vary: 39% (Heyse) to 100% (Lam)
- Younger age group (≤13 years) showed greater pain improvement (p=0.02)
- Caution: 1 in 4 children advance their Kellgren-Lawrence score during treatment [66]
→ Kellgren-Lawrence = arthritis grading scale (0-4); higher = worse arthritis
Best for: Non-displaced Grade I-II lesions, skeletally immature patients, patients with minimal symptoms. Patients who receive nonoperative management seldom recover to their previous level of sports activity [10].
2. Hyaluronic Acid (HA) and PRP Injections
HA Systematic Review Evidence (RCTs)
→ HA is a gel-like lubricant naturally in joints. PRP uses your own blood's healing factors. Neither creates new cartilage, but they may reduce inflammation and pain.
A systematic review of 3 randomized controlled trials (132 patients) found HA injection as an adjunct to microfracture provides clinically important improvements [15]:
→ RCT = Randomized Controlled Trial (gold-standard study design); adjunct = added alongside another treatment
- AOFAS scores: Greater improvement vs microfracture alone (moderate effect size, p=0.02)
→ AOFAS = American Orthopaedic Foot & Ankle Society score (0-100); measures pain, function, alignment
- VAS-pain scores: Significantly greater improvement (very large effect size, p<0.001)
- After failed surgery: HA injections significantly improved clinical scores
PRP Meta-Analysis (December 2024)
A 2024 meta-analysis from the Journal of Orthopaedic Surgery and Research (5 RCTs) found [67]:
8.66
AOFAS improvement in talar cartilage injuries
[67]
-0.62
VAS pain reduction (SMD)
[67]
→ SMD = Standardized Mean Difference; negative value = less pain (good); OA = osteoarthritis
- Subgroup analysis: More significant effect in patients with talar cartilage injuries specifically
- PRP treatment effect remained effective in long-term follow-up
- Greater advantage for cartilage injuries of the talus vs general ankle OA
PRP Complications (2025 Systematic Review)
A 2025 systematic review from Arthroscopy Journal found important safety data [68]:
- 674 patients receiving PRP vs 749 receiving alternatives
- Complication rate: 41.1% (PRP) vs 33.7% (comparison group)
- Most common: treatment-site pain (15.1% vs 10.2%, p<0.01)
- Number needed to harm: 13 patients
→ NNH = on average, for every 13 patients treated with PRP, 1 extra complication occurs vs alternative
Network Meta-Analysis of Adjunctive Therapies (2024)
A network meta-analysis of 6 RCTs (295 patients) compared adjuncts to microfracture [69]:
→ Network meta-analysis = combines multiple studies to compare treatments that may not have been directly compared
- Compared: PRP, HA, collagen scaffold, pulsed electromagnetic fields (PEMF)
→ PEMF = therapy using magnetic waves to stimulate healing
- PRP + MF showed superior final VAS and AOFAS scores vs MF only (p<0.01)
→ MF = microfracture
FDA Note
HA was FDA approved in 1997 for knee osteoarthritis only. FDA has not approved intra-articular HA for ankle joints, though it is commonly used off-label [17]. A 2024 CADTH rapid review confirmed limited evidence for ankle applications [70].
→ Intra-articular = injected into the joint; off-label = approved for another use but legally prescribed by doctors for this one
3. Bone Marrow Stimulation (Microfracture/Drilling)
Long-Term Survival Data (2024-2025)
→ Microfracture pokes tiny holes in bone to cause bleeding. Stem cells from bone marrow form a clot that becomes "fibrocartilage" - scar-like tissue that's not as strong as real cartilage, but can reduce pain. Like patching with duct tape instead of the original material.
82%
10-year survival (JBJS 2024)
[22]
82%
15-year survival (same study)
[22]
87-100%
Survival at 5+ years (2025 review)
[71]
61%
Revision/secondary OLT success
[21]
2025 Systematic Review: Mid-to-Long Term Outcomes
A 2025 systematic review of 43 studies (2,721 patients, 5-24 year follow-up) found [71]:
- BMS is the most frequently used treatment for small (<1.5 cm²) primary lesions
→ BMS = Bone Marrow Stimulation; primary = first-time lesion (not a recurrence)
- Survival rates (no revision) at latest follow-up: 87.4% to 100%
- Primary lesions: 82% success rate (95% CI: 78-86%)
2025 Five-Year Follow-Up Study
A 2025 study in Indian Journal of Orthopaedics reports 5-year outcomes [72]:
- Short-term efficacy well-established; mid-to-long term outcomes remain ambiguous
- Focus on pain and functional recovery at 5 years
DGOU 2024 Guidelines
→ DGOU = German Society of Orthopedics and Traumatology (respected European guidelines)
- Debridement with BMS supported for lesions <1.0 cm² without bony defect [24]
→ Debridement = surgically cleaning out damaged/dead tissue
- For lesions >1.0 cm², additional scaffold is recommended [24]
→ Scaffold = a supportive framework (like construction scaffolding) that helps new tissue grow
- Consensus: OCLs >1.5 cm² require membrane scaffold augmentation [24]
→ OCL = Osteochondral Lesion; augmentation = adding something to enhance the procedure
Cystic Lesions (2025 Meta-Analysis)
A 2025 meta-analysis found no clinical difference between simple and cystic lesions if cyst depth <5-6mm, medial talus location, and patient <40 years old [26].
→ Cystic lesion = has a fluid-filled hole in the bone; simple = just cartilage damage without a bone cyst
However, for large cystic OLT (>300 mm³), autologous osteochondral transfer showed superior survival vs BMS (p=0.042) [27].
→ Autologous = using tissue from your own body (from another location)
2026 CARTILAGE Meta-Analysis: BMS Produces Inferior Cartilage
A 2026 meta-analysis of second-look arthroscopy studies found BMS produces significantly inferior cartilage quality compared to all other techniques [169]:
53%
BMS cartilage quality success
94%
Osteochondral transplant success
81%
Cartilage implantation success
→ When surgeons go back in and look at the repair tissue, BMS cartilage is visually and structurally worse than what other techniques produce. This may shift first-line treatment away from microfracture.
2026: Immediate Weightbearing After BMS Appears Safe
A matched cohort study found no significant difference in outcomes between immediate and delayed weightbearing after arthroscopic BMS at 12 months [170]. This may simplify post-op protocols.
Long-term Concerns
Fibrocartilaginous repair tissue tends to degenerate over time. Good clinical outcomes at midterm, but radiological outcomes showed repair tissue surface damage [20]. This may be a harbinger for long-term problems [28].
→ Fibrocartilaginous = scar-like cartilage (weaker than original hyaline cartilage); harbinger = warning sign
4. BioCartilage / Scaffolds / AMIC
What It Is
→ BioCartilage and AMIC add a "helper layer" on top of microfracture. A scaffold (like a cartilage-protein sponge) gives healing cells a structure to grow on. Like providing scaffolding for construction workers instead of building in mid-air.
BioCartilage is dehydrated, micronized allogeneic cartilage containing extracellular matrix (type II collagen, proteoglycans, cartilaginous growth factors). It acts as a scaffold over microfractured defects [29].
→ Allogeneic = from a donor (not your own tissue); extracellular matrix = the "glue" between cells; proteoglycans = cushioning molecules in cartilage
Systematic Review Outcomes
87.5%
Complete defect infill
[30]
28%
Reoperation (MF alone)
[30]
AMIC 2024 Systematic Review and Meta-Analysis
A June 2024 systematic review of 15 studies (12 case series for meta-analysis) [31]:
→ AMIC = Autologous Matrix-Induced Chondrogenesis; combines microfracture with a collagen membrane
- VAS, AOFAS ankle-hindfoot, and Tegner scores at last follow-up showed statistically significant improvement (p<0.001)
→ Tegner score = activity level scale (0-10); higher = more active lifestyle
- VAS improved 4.45-4.6 points from baseline at 1-5 year follow-up
AMIC 10-Year Cohort Study (2025)
A 2025 study in Cartilage evaluated 10-year outcomes [73]:
- "Gold standard surgical treatment for OCLs of the talus still needs to be established"
- AMIC is a commonly applied 1-stage procedure with good short/mid-term results
→ 1-stage = done in a single surgery (vs 2-stage which requires harvesting cells, growing them, then implanting)
- 10-year cohort tracked FFI-D scores preop and at 1, 5, and 10 years
→ FFI-D = Foot Function Index (German version); measures pain and disability
Arthroscopic AMIC 5-Year Follow-Up (2026 CARTILAGE)
Efrima et al. evaluated arthroscopic AMIC at 24 and 60 months [74]:
- Significant clinical improvement maintained at 5 years; improvement peaks in first 2 years then plateaus
- Patients older than 33 had significantly worse outcomes
- Increased age and BMI associated with inferior results
- Arthroscopic approach shows promise for reduced morbidity
DGOU 2024: Scaffold Sizing Guidance
The German cartilage society guidelines now offer clear sizing thresholds [128]:
- BMS alone: only for lesions <1.0 cm² without bony defect
- Scaffold augmentation recommended for lesions >1.0 cm² and for cystic defects
- Warning: bone grafting alone has a 46% revision rate — scaffolds improve this significantly
- Arthroscopy has technical limits for radical debridement of extended cystic lesions
2024 EMCA/BioCartilage Systematic Review
A 2024 systematic review of 162 ankles across 5 studies found [29]:
- Subjective improvement and some MRI advantages over BMS alone
- However, no consensus on EMCA's precise role — longer comparative studies needed
Concerns
- High reoperation rate: 54.6% underwent subsequent surgery (mostly hardware removal) [33]
- Only 14% of revisions were graft-related complications [33]
- MaioRegen scaffold: 31% failure rate (5/16), 4 required prostheses [34]
For revision cases: This branch looks strongest when the surgeon treats it as a lower-morbidity bridge or bone-restoring/staging step, not as a definitive standalone answer for failed grafts.
5. Osteochondral Autograft (OATS/Mosaicplasty)
Long-Term Outcomes (10+ Years)
→ OATS takes a plug of healthy cartilage+bone from your knee and presses it into the ankle defect like a cork in a bottle. Your own tissue = no rejection risk. Downside: creates a new wound in your knee.
Systematic review with 10.2 year mean follow-up (610 patients, 10 studies) [35]:
- Overall success rate: 72% at long-term follow-up [35]
- Mean AOFAS improved from 60.4 to 86.2 at minimum 10-year follow-up [36]
- VAS decreased from 6.3 to 2.0 [36]
- Results do not deteriorate over time [37]
2024 Systematic Review Update
A 2024 systematic review in JOSR evaluated OATS outcomes [75]:
- OATS remains the gold standard for large cystic OLT (>150 mm²)
- Return to sport rate: 76-83% at 2-year follow-up
- Superior to BMS for lesions >1.5 cm² with bone involvement
2024 Ten-Year AOT Review
A 2024 study with minimum 10-year follow-up reported strong long-term durability [151]:
- Mean lesion size: 122.3 mm², 43.6% cystic lesions, 28.2% had prior microfracture
- FAOS improved from 51.9 to 75.3, VAS from 6.6 to 1.6
- Only 2 failures (5.1%) — both had prior microfracture plus postoperative cysts
- 94.9% survival rate at minimum 10-year follow-up
2026 Multi-Plug AOT for Large Lesions
A 2026 series using multiple plugs for large OLTs [152]:
63.8→97.4
AOFAS at 12 months
0%
Graft failures / infections
- No conversion to arthrodesis or arthroplasty in first year
- However, knee donor-site pain across reviews still ranges 6.7%-16.9%
Autograft vs Allograft (Meta-analysis of 1174 procedures)
Autografts significantly outperform allografts [38]:
→ Autograft = your own tissue; Allograft = donor tissue (like a transplant)
- Lower revision surgery rate: OR 7.2 (p<0.0001)
→ OR = Odds Ratio; 7.2 means allografts are 7.2x more likely to need revision surgery
- Lower failure rate: OR 5.1 (p<0.0001)
2024-2025 Updates on Surgical Technique
Recent advances in osteochondral autograft procedures [76]:
- Novel arthroscopic-assisted techniques reduce surgical morbidity
- CT/fluoroscopic navigation improves plug placement accuracy
- Single-plug technique preferred for lesions <15mm diameter [77]
Donor Site Morbidity (Major Concern)
→ Donor site morbidity = problems caused at the site where tissue was taken (the knee, in OATS procedures)
Systematic review of 1726 patients (21 studies) [39]:
16.9%
Knee-to-ankle morbidity
[39]
5.9%
Knee-to-knee morbidity
[39]
44%
Pain/instability (ankle MCP)
[39]
→ MCP = mosaicplasty (multiple small plugs); DSM = donor site morbidity
2025 Donor Site Study
A 2025 study on predictors of donor site morbidity found [40]:
- Overall DSM rate: 9.8% using Lysholm Knee Score
- Higher BMI significantly correlated with increased morbidity
- Number of plugs harvested is key predictor
Ipsilateral Talus as Donor Site (2024)
Emerging technique using autologous ipsilateral talus grafts [78]:
→ Ipsilateral = same side; taking cartilage from another part of the SAME ankle instead of the knee
- Eliminates knee donor site morbidity entirely
- Comparable biomechanical properties to defect site
- Limited to smaller lesions due to available donor area
Key issue: Patients receiving 2+ plug grafts have worse knee scores (96 vs 98, p<0.05). Knee and talus cartilage do not biomechanically match [39].
6. Osteochondral Allograft (Fresh Talar Allograft)
Survival Rates (Systematic Review of 522 ankles)
→ Uses cartilage+bone from a deceased donor (cadaver) instead of your own body. Like an organ transplant for cartilage. Advantage: no donor site pain. Downside: potential rejection, tissue isn't "alive" like yours.
86.6%
Aggregate graft survival
[41]
2024 Systematic Review Update
A 2024 comprehensive review of fresh osteochondral allograft (OCA) outcomes [79]:
- Fresh OCA has become the preferred treatment for large OLT (>150 mm²)
→ OCA = Osteochondral Allograft (bone + cartilage from a donor)
- Graft viability critical: optimal implantation within 28 days of harvest
→ Viability = cells are still alive; fresh grafts need to be implanted quickly before cells die
- 5-year survival rates: 85-92% when proper tissue banking protocols followed
2025 Fresh Allograft Outcomes Study
Recent 2025 data on fresh talar allografts [80]:
88%
5-year survival (fresh grafts)
14%
Revision rate at 5 years
Long-Term Data (Gross et al.)
- 12-year mean follow-up: 6/9 grafts remained viable [43]
- 3 patients required fusion due to graft resorption/fragmentation [43]
- Success rates range from 73-100% across studies [43]
Bulk vs Shell Allograft Comparison (2024)
A 2024 comparative analysis evaluated bulk hemitalar vs shell allografts [81]:
- Shell grafts: Better incorporation rates, lower revision rates
- Bulk hemitalar: Reserved for massive defects (>50% talar dome)
- Surgeon experience significantly impacts outcomes
Revision Options After Failed Allograft
Systematic review of 115 failed bulk allografts [44]:
- Ankle arthrodesis: 34 cases (most common)
- Revision allograft: 30 cases
- Total ankle arthroplasty: 20 cases
- Mean time to revision: 53.3 months
2024 Fresh OCA Long-Term Follow-Up (9.2 Years)
A 2024 study with mean 9.2-year follow-up provided the strongest recent supportive data [153]:
94.3%
Graft survival at 5 years
85.3%
Graft survival at 10 years
66.7%
Returned to sports/recreation
16.7%
Required further surgery
- 50% were still participating in sports at latest follow-up
- Concluded fresh OCA is reasonable for young active patients who failed prior surgery or have massive defects
2024-2025 Revision Allograft Outcomes
New data on revision allograft surgery [82]:
- Revision OCA has lower success rates than primary (68% vs 87%)
- Prior surgery type affects revision outcomes
- Failed BMS prior to OCA does not negatively impact OCA success
2024 Pooled Revision Evidence
The 2024 non-primary OLT systematic review (50 studies, 806 ankles) found less favorable pooled results for OCA as a revision procedure [148]:
- OCA did not significantly improve pooled AOFAS or pain outcomes in revision cases
- Clinical failures occurred in 14 of 72 OCA cases (19%)
- OCA and HemiCAP looked less effective as revision procedures than ACI or OAT
Critical for Raymond: Allografts have 5-7x higher failure/revision rates vs autografts [38]. A first graft lasting 12 years is a meaningful personal data point, but repeat allograft needs a case-specific reason to beat osteoperiosteal revision or OATS [45].
7. Particulated Juvenile Cartilage Allograft (DeNovo)
Systematic Review Evidence (241 patients, 2024)
→ DeNovo uses tiny cartilage pieces from young donors (under 13). Young cartilage has more "stem cell-like" properties. Mixed with fibrin glue and placed into the defect.
9.8 mo
Mean time to failure
[46]
Key Findings
- AOFAS improved from 58.5 to 83.9 [46]
- Most common complication: allograft hypertrophy (13.2%) [46]
- Lesions >125mm² had significantly increased failure risk [46]
- Male sex was a risk factor; age, BMI, prior surgery were NOT [46]
2024-2025 Clinical Updates
Recent studies have provided more data on particulated juvenile cartilage [83]:
- 5-year follow-up shows continued concern about durability
- Tissue quality on MRI remains fibrocartilaginous rather than hyaline
- Newer scaffold-assisted techniques may improve outcomes
11-Year Case Report
Long-term case report with second-look arthroscopy showed graft survival at 11 years [47].
Product Availability Note (2024)
DeNovo NT (natural tissue) is manufactured by Zimmer Biomet. Availability may be limited in some regions, and cost remains significant (~$5,000-8,000 for the graft alone) [84].
Conclusion: High complication and failure rates call into question efficacy for large OLTs. Cannot be currently recommended for lesions >125mm² [46].
8. Subchondralplasty (Bone Substitute Injection)
What It Is
→ Bone under cartilage has a hole (cyst) filled with fluid. Subchondralplasty injects bone cement-like paste through a needle to fill it. The paste hardens and becomes part of your bone. Like filling a pothole in a road.
Minimally invasive technique injecting calcium phosphite paste into subchondral cysts. The paste crystallizes like cancellous bone to fill the defect [48].
Reported Outcomes
- 6-month CT: >50% of cyst replaced by bone substitute integrated with trabecular network [49]
- 1-year follow-up: Pain-free walking, preserved ROM [49]
- Patients report little to no post-op pain, same-day discharge [50]
2024-2025 Subchondralplasty Updates
Recent studies have expanded understanding of this technique [85]:
- 2024 systematic review: 78% patient satisfaction at 2-year follow-up
- Best outcomes in isolated bone marrow edema without full-thickness cartilage loss
- May be combined with microfracture or scaffolds for comprehensive treatment
Calcium Phosphate vs Other Substitutes (2024)
A 2024 comparative study examined different bone substitutes [50]:
- Calcium phosphate (AccuFill): Most commonly used, good integration
- Bioresorbable materials show promise but lack long-term data
- Injectable DBM (demineralized bone matrix) alternatives emerging
Complications
- Traditional bone-grafting has 27-63% cyst recurrence rate [48]
- Reported complications: AVN of talus, osteomyelitis (rare) [51]
- Overfilling can cause pain; intra-articular extravasation is a risk [49]
Retrograde Drilling + Bone Graft
Alternative approach: retrograde drilling has ~90% success rate for intact cartilage lesions [52]. However, failure rates can reach 20% due to inaccurate targeting [53].
Navigation-Assisted Drilling (2024-2025)
Newer navigation technologies improve accuracy [52]:
- 3D CT-based navigation: 95% accuracy in cyst targeting
- Fluoroscopic guidance: Traditional but less precise
- Patient-specific instruments emerging for complex cases
For Raymond: Dr. Choung noted the cysts are small and mostly inflammatory, making subchondralplasty not currently indicated.
9. Autologous Chondrocyte Implantation (ACI/MACI)
20-Year Follow-Up Data (2024)
→ ACI: Two surgeries. First, surgeon takes healthy cartilage cells and a lab grows millions more (4-6 weeks). Second surgery implants them. MACI grows cells on a scaffold membrane for easier implantation.
A landmark 2024 study evaluated first-generation ACI with minimum 20-year follow-up [90]:
- AOFAS improved from 40.4 to 82.7 at final follow-up (p<0.0005)
- Pain NRS improved from 7.8 to 4.8 (p<0.0005)
- All patients preserved their ankle joints without needing major revision surgery
- Some decline in outcomes and activity restrictions reported over time
MACI Systematic Review (2024)
A 2024 systematic review of 166 MACI patients [91]:
78-95
Post-op AOFAS range
89%
Success rate (meta-analysis)
Indications
- MACI recommended for defects >2 cm² [24]
- Requires two surgeries (biopsy + implantation)
- Higher cost due to cell culture process
- Best for larger lesions where BMS/scaffolds are insufficient
Revision Context (2024)
The 2024 revision systematic review found ACI had the best pooled PRO performance in non-primary OLTs, but the dataset was tiny (only 7 ankles) [148]. DGOU 2024 also notes there is no evidence of added benefit of ACI over acellular scaffolds in the talus [128].
For Raymond: ACI/MACI is more compelling when the problem is primarily cartilaginous. A bone-supported graft wearing out points more toward osteochondral or osteoperiosteal reconstruction.
10. Stem Cell & Regenerative Therapies (Emerging)
DGOU 2024 Position
→ Experimental treatments using stem cells (from fat, bone marrow, etc.) to regenerate cartilage. Stem cells are "blank" cells that can become cartilage cells. Exosomes are tiny healing signal packets cells release.
The German Society of Orthopedics and Traumatology considers adipose-derived MSC therapy for OLT experimental [92]:
→ Adipose-derived = from fat tissue; MSC = Mesenchymal Stem Cells (cells that can become cartilage)
- Limited literature to support adipose-derived MSC use in OLT
- Regulatory restrictions vary by country
- More research needed before routine clinical use
Recent Case Studies (2025)
A November 2025 case using heparin-conjugated fibrin hydrogel with MSCs [93]:
→ Hydrogel = gel-like scaffold that holds cells in place; fibrin = natural clotting protein
- VAS decreased from 60 to 40 at 12 months
- AOFAS increased from 69 to 77
- MRI showed progressive cartilage regeneration with near-complete defect filling
Exosome Therapy (Pre-Clinical)
Exosomes are emerging as potential therapy for cartilage repair [94]:
- MSC-derived exosomes promote chondrocyte proliferation
→ Exosomes = tiny particles cells release to communicate; chondrocytes = cartilage cells; proliferation = growth/multiplication
- Anti-inflammatory and pro-regenerative properties
- Injectable hydrogel delivery systems under development (2025)
- Clinical trials in OLT specifically still needed
CBMA (Concentrated Bone Marrow Aspirate)
CBMA is a simpler stem cell approach harvested during surgery [1]:
→ CBMA = liquid from inside your bones (rich in stem cells) concentrated and applied to the defect
- Single-stage procedure (no cell culture needed)
- Often combined with microfracture or scaffolds
- Technique recommended for lesions >1.5 cm² with bone defects <5mm
Status: These therapies show promise but remain largely experimental. No large RCTs yet for ankle OLT specifically.
11. Extracorporeal Shockwave Therapy (ESWT)
2025 Study: ESWT + Microfracture + HA
→ High-energy sound waves (tiny sonic booms) directed at the damaged area. Stimulates healing by increasing blood flow and reducing inflammation. Non-invasive, done in office visits. Like jump-starting your body's healing response.
A July 2025 study evaluated combined therapy for OLT [95]:
7.2→2.1
VAS pain reduction
- ESWT synergizes with HA injections to reduce post-microfracture pain
- Promotes early joint function recovery
- Anti-inflammatory, pro-angiogenic, and analgesic effects
2024 Research: Mechanism of Action
A 2024 study in Bone & Joint Research [96]:
- ESWT reduced fibrosis and promoted hyaline cartilage formation
- Increased type II collagen (good) and decreased type I collagen (bad)
- Enhanced TGF-β/BMP signaling for cartilage regeneration
- Stimulated subchondral bone regeneration in a bottom-to-top pattern
ESWT vs PRP After Microfracture
A 2023 study comparing MF+ESWT vs MF+PRP [97]:
- MF+ESWT had higher AOFAS scores at 2-year follow-up
- Lower T2 mapping values (indicating more hyaline-like cartilage)
- Better cartilage quality on MRI assessment
ISMST Recognition
OLT has been recognized as an indication for ESWT by the International Society for Medical Shockwave Treatment since 2008 [98].
Best for: Non-invasive adjunct therapy, especially combined with microfracture. Can reduce pain and improve outcomes without additional surgery.
12. Pulsed Electromagnetic Field Therapy (PEMF)
2024-2025 Research Evidence
→ PEMF devices generate low-frequency electromagnetic waves that penetrate tissue. Thought to stimulate cellular repair, reduce inflammation. Completely non-invasive - just place device near ankle daily. Like giving cells a gentle energy boost.
Recent studies show promising mechanisms for PEMF in cartilage repair [99]:
- Inhibits cartilage degradation and reduces pro-inflammatory factors
- Increases expression of chondrogenic markers (COL2, ACAN) [100]
- Activates Sirt1 pathway to block inflammatory factors [101]
- Promotes mesenchymal stem cell chondrogenesis via calcium signaling
NASA Research
NASA Johnson Space Center developed a PEMF device showing that waveform variation influences genetic regulation of chondrocytes from early OA patients [102].
Limitations
- Cannot reverse advanced arthritis or regenerate destroyed cartilage
- May slow progression and manage symptoms
- Most research on knee OA, limited ankle-specific studies
- Home devices vary in quality and efficacy
Status: Promising adjunctive therapy, but not a standalone treatment for OLT. Best as part of a comprehensive conservative management approach.
13. Physical Therapy & Rehabilitation
2024 Systematic Review: Protocol Variation
→ PT isn't just for after surgery - it's core treatment at every stage. Focuses on reducing pain, improving motion, strengthening muscles, and training ankle to move properly (proprioception).
A 2024 meta-analysis of 200+ studies found large variation in rehab protocols [103]:
- No universally standardized post-operative protocol exists
- Collaboration between PT and surgeon is key for outcomes
- Patient perspectives often underreported in protocols
Conservative Treatment PT Components
Key elements of physical therapy for OLT [104]:
- Peroneal strengthening: Critical for ankle stability
- Proprioceptive training: Balance boards, single-leg exercises
- Range of motion: Maintain flexibility without aggravating lesion
- Activity modification: Avoid high-impact activities
Post-Surgical Rehabilitation Timeline
General rehab phases after cartilage surgery [105]:
- Phase 1 (0-6 weeks): Protected weight bearing, gentle ROM, control swelling
- Phase 2 (6-12 weeks): Progressive weight bearing, strengthening begins
- Phase 3 (3-6 months): Full weight bearing, agility training
- Phase 4 (6-12 months): Return to sport, high-impact activities
2025 Research Gap
A 2025 narrative review noted that high-quality studies on physical therapy for OLT remain relatively few [106]. However, preoperative assessment and postoperative functional rehabilitation are considered critical components of successful OLT management.
Key point: Finding a PT experienced with ankle cartilage injuries can make a significant difference in outcomes. Full recovery often takes up to 12 months.
End-Stage Options: Fusion vs. Replacement
→ When cartilage treatments fail: Fusion (arthrodesis) permanently joins ankle bones = eliminates pain AND motion. Replacement (arthroplasty) = artificial joint like hip/knee, preserves motion but needs eventual revision. Both are major surgeries with long recovery.
Total Ankle Arthroplasty (TAA) vs. Ankle Fusion
Meta-Analysis Summary (4,312 TAA, 1,091 fusion procedures)
- No statistically significant difference in clinical outcome, patient satisfaction, or survival [54]
- TAA has better short-term PROMs and gait [55]
- In long-term, fusion favored due to lower complications/revisions [54]
2024-2025 TAA Advances
Recent studies show significant improvements in newer implant designs [86]:
- Fourth-generation implants (INFINITY, INBONE II): Lower revision rates than older designs
- 3D-printed custom implants emerging for complex revision cases [87]
- Patient-specific alignment guides improve surgical accuracy
TAA in Young Patients (<50-55 years)
1.8-2.65x
Higher revision risk
[56]
91.8%
10-yr implant survival
[58]
Third-generation designs show improved results vs older prostheses. At medium-term, TAA is at least as effective in patients <50 as in older patients [59].
2025 National Registry Data
Recent national registry analyses provide large-scale outcome data [88]:
- 5-year revision rate: 8-12% across major registries
- Age <55 consistently identified as revision risk factor
- High-volume surgeons (>20 cases/year) have better outcomes
2026 Landmark: 25-Year, 41,000-Patient Registry (England)
The largest-ever comparison of TAR vs ankle fusion (10,335 TARs vs 30,704 fusions, 1998-2023) [172]:
2.73x
TAR revision hazard vs fusion
35%
TAR 25-year intervention-free survival
69.2%
Fusion 25-year intervention-free survival
- TAR revision rates: 6.1% at 5y, 10.2% at 10y, 13.55% at 20y
- Fusion revision rates: 2% at 5y, 2.5% at 10y, 3.1% at 20y
- Myth debunked: Adjacent joint fusion was NOT higher after ankle fusion (25-year rates: AF 8.64% vs TAR 6.82%, not significant)
→ The common argument that fusion causes adjacent joint damage may not hold up. This 25-year data suggests fusion is actually more durable than replacement, though it sacrifices motion.
Ankle Fusion Long-Term Outcomes
- Fusion rate: 80-100% in most studies [60]
- Adjacent joint arthritis: 10-60% rate long-term [60]
- Subtalar arthritis progression: 36.6% [61]
- Function deteriorates over time [60]
2024-2025 Arthrodesis Techniques
Newer fusion techniques show improved outcomes [89]:
- Arthroscopic fusion: Faster recovery, less wound complications
- Intramedullary nail fixation: Higher union rates for complex cases
- Biologics (BMP-2, PRP): May enhance fusion rates in high-risk patients
Conversion: Fusion to TAA
For painful fusions, conversion to TAA is possible [62]:
- VAS improved from 7.8 to 2.5; AOFAS from 32 to 72.4
- Salvage tibiotalocalcaneal arthrodesis: 2.3%
- Absent fibula is absolute contraindication
Key point: All doctors agreed ankle fusion should be avoided for someone young with good ROM. Cleveland Clinic noted TAA would be a later escalation step.
Total Talar Replacement (TTR)
Total talar replacement is a newer procedure where the entire talus bone is removed and replaced with a custom 3D-printed prosthetic implant. Because the talus has no direct muscle/tendon attachments and limited blood supply, it is uniquely suitable for full bone replacement.
→ Unlike total ankle replacement (TAR) which replaces the joint surfaces of both the tibia and talus, TTR replaces only the talus bone itself. TTR is indicated when the talus is too damaged for cartilage repair (AVN, collapse, large cystic defects unresponsive to treatment).
The Procedure
- CT scans of both ankles are taken; the healthy side is mirrored to design the implant [158]
- Implant is custom 3D-printed in cobalt-chromium, alumina ceramic, or pure titanium
- Three sizes manufactured (90%, 95%, 100% of native bone volume) for intraoperative trial fitting
- Rehab: splinting 2 weeks, progressive weight-bearing weeks 2-4, full weight-bearing weeks 4-6, normal activities by week 12
FDA Approvals
- 2021: Additive Orthopaedics talus spacer — first-ever FDA HDE for a 3D-printed talus implant (cobalt-chromium) [159]
- November 2023: restor3d Total Talus Replacement — FDA HDE approval. Device survivorship: 96.3%, reoperation rate: 14.8% [160]
- December 2024: 4WEB Medical Talar Replacement Device — FDA HDE approval based on 30-patient retrospective study [161]
Outcomes Data
Systematic review of 22 studies (191 patients, 196 tali) [162]:
94.5%
Implant survivorship
[159]
69.6%
Significant functional improvement
[162]
0.1%
Cumulative implant failure rate
[162]
8.2 → 2.1
VAS pain score (titanium series)
[163]
Additional clinical results:
- Cobalt-chrome series (38 patients, 2025): VAS improved from 8.43 to 2.67, AOFAS from 36.33 to 81.78 (p<0.001) at mean 22.1 months [164]
- Pure titanium series (62 patients, 2025): First clinical series using commercially pure titanium. VAS 8.2 → 2.1, only 2 cases of mild implant settling [163]
- Ceramic implants (19 patients, Japan): Median 12.7-year follow-up — JSSF score 97/100, zero failures requiring revision [165]
- Multicenter safety (15 patients, 2024): 93% survivorship at mean 25.9 months. 33% adverse event rate, but 0 were device-related [166]
TTR vs. Ankle Fusion vs. TAR
- vs. Fusion: TTR preserves tibiotalar and subtalar motion; fusion eliminates ~70% of sagittal plane mobility. TTR preserves leg length. If TTR fails, fusion remains a salvage option [54]
- vs. TAR: TAR replaces joint surfaces of both tibia and talus; TTR replaces only the talus. TAR 10-year survival: 74-84%. TTR data is shorter-term but 93-96% survivorship. Combined TTR+TAR is possible if both are needed [164]
Longevity: The "Every 10 Years" Question
Dr. Salk stated the implant would need replacing approximately every 10 years. What the data shows:
- Longest data (Japanese ceramic): 85% still in place at 10-36 years. Zero revisions needed at median 12.7-year follow-up [165]
- Modern 3D-printed implants: Maximum follow-up is only ~3-4 years. No 10-year data exists for current cobalt-chrome or titanium designs
- TAR comparison: Modern TAR 10-year survival 74-84%, 15-year survival 63%, 20-year survival 58%. Most surgeons tell patients under 70 to expect 10-15 years
- Key concern: Adjacent joint osteoarthritis is progressive — in Morita's 10+ year follow-up, degenerative tibial changes appeared in 90% of cases (though none required revision) [165]
→ The "10 years" estimate is likely borrowed from TAR data. The only long-term TTR data (ceramic) actually suggests longer survival, but modern 3D-printed implants are too new to know for certain.
Risks and Complications
- Complication range across studies: 0% to 33% [162]
- Most common long-term issue: Adjacent joint osteoarthritis (tibial plafond in 90%, navicular 16%, calcaneus 11%) [165]
- Implant settling/subsidence (2 cases in titanium series, associated with insufficient subchondral bone support)
- If it fails: primary salvage is tibiotalocalcaneal (TTC) fusion; revision TTR or adding TAR component also possible [167]
- Warning from revision data: Nearly 1/3 of talar revisions go on to a second revision [168]
Who Is It For?
FDA-approved indications include [160]:
- Avascular necrosis of the talus (with or without collapse, cysts, or non-union)
- Large, uncontained, unstable, or cystic talar osteochondral defects with risk of collapse
- Talar osteochondral defects not responsive to traditional treatments
- Non-union following talar fracture or extrusion unresponsive to conservative treatment
For Raymond: TTR is not the first-line treatment for the current OLT. The standard treatment algorithm places this lesion in the osteochondral allograft/autograft/MACI category. TTR is reserved for when the talus is "unreconstructable" — end-stage AVN or complete talar collapse. However, if cystic changes progress to AVN, TTR becomes a viable option. The FDA indications specifically include "large, uncontained, unstable, or cystic talar osteochondral defects with risk of collapse." At age 29, the key advantage over fusion is motion preservation; the key risk is that if it fails, salvage options become progressively more limited. The technology is advancing rapidly (three FDA approvals in 3 years, new materials in 2025), so by the time TTR might be needed, implants may be significantly improved.
Lifestyle Factors & Supplements
14. Body Weight and BMI Impact
2025 German Cartilage Registry Data
→ Extra weight = more stress on ankle cartilage with every step. Obesity linked to worse surgical outcomes. Like driving on a weak tire - heavier load = faster wear. Weight management can significantly affect how well treatments work.
A 2025 study from the German Cartilage Registry (303 patients) examined BMI impact on OLT surgical outcomes [107]:
- Both normal weight (BMI <30) and obese (BMI ≥30) groups showed significant improvement
- Higher BMI associated with higher prevalence of ankle sprains and OCTs
- Obesity incidence is rapidly increasing, making this clinically important
2024 JBJS Long-Term Survival
The 10-year survival study analyzed BMI as a risk factor for treatment failure [22]:
- BMI ≥30 kg/m² was included as a prognostic factor
- 82% 10-year survival overall, with obesity as potential modifier
Takeaway: While not a contraindication to surgery, maintaining healthy weight optimizes outcomes.
15. Nutritional Supplements
2025 Systematic Review
→ Glucosamine, chondroitin, collagen = cartilage building blocks taken as pills/powders. Gives body extra raw materials. Research is mixed - may help pain modestly. Generally safe but unlikely to regenerate destroyed cartilage.
A comprehensive 2025 review of 146 studies on glucosamine and chondroitin [108]:
- Over 90% of efficacy studies reported positive outcomes
- Most safety studies indicated minimal or no adverse effects
- Type II collagen peptides showed consistent efficacy
- Often combined with MSM, curcuminoids, and HA
2025 RCT Results
A 12-week randomized controlled trial (52 participants) on combination supplements [109]:
25%
Sport/recreation improvement
28%
Quality of life improvement
Important Caveats
NIH/NCCIH notes mixed evidence [110]:
- Studies have had inconsistent results
- Expert evaluations have reached conflicting conclusions
- A 2024 meta-analysis found glucosamine/chondroitin did not significantly reduce joint pain or prevent joint space narrowing [111]
Bottom line: Generally safe to try for 3-6 months. May provide modest pain relief. Not a substitute for proper medical treatment.
16. Gene Therapy (Future Direction)
Current Status
→ Gene therapy = giving cells new instructions. Delivers genes that tell cells to produce more cartilage proteins. Still experimental for ankle - most research in lab animals - but represents the future of repair.
Gene therapy for cartilage is advancing but not yet clinically available for ankle OLT [112]:
- Most trials use MSCs (36%), followed by PRP (20%) [113]
- Recombinant AAV vectors delivering FGF-2, IGF-1, SOX9 show promise in animal models
- Allogeneic transduced chondrocytes in early clinical trials for OA
Mayo Clinic Phase 1 Trial
A recycled cartilage auto/allo implantation technique is being studied [114]:
- Combines 10-20% autologous chondrons with 80-90% allogenic MSCs
- Under FDA IND status at Mayo Clinic
- Represents future direction for cartilage repair
Status: Experimental. Not available for routine clinical use in 2025.
17. Return to Sport: What to Expect
Return to Sport Rates by Procedure
→ Getting back to sports is a long journey. Procedure type determines wait time. Simpler = faster return but less durable tissue. Complex = longer wait but better long-term tissue. Most return to some sports, but often modify activities.
Systematic review of 2,347 OLT cases [115]:
76%
Continue sports long-term
73%
Return to pre-injury level
Timeline by Procedure Type
- Microfracture: ~4 months return (ideal for athletes) [116]
- AMIC/BMDCT: ~12 months for full return [117]
- Osteochondral transplant: 6-12 months depending on complexity
2024 Osteochondral Allograft Data
A 2024 study on OCA transplantation in athletes [118]:
- 67% returned to sports/recreational activities after OCA
- 74% of competitive athletes returned to sport
- 50% still participating at latest follow-up
2024 Pediatric Microfracture Data
For skeletally immature athletes with OLT [119]:
- 100% returned to sports in pediatric cases
- Good clinical and MRI outcomes
- Microfracture effective for lesions <10mm in children
Activity Modification
Important considerations from the literature:
- Many patients reduce high-impact and contact sports post-surgery
- Lower-impact activities (swimming, cycling) may be safer long-term
- Return to sport level often doesn't match pre-injury regardless of procedure
18. Surgical Approaches: Malleolar Osteotomy (2024-2025)
→ To access OLT, surgeons sometimes need to temporarily cut the ankle bone (malleolus) and move it aside. Like opening a hatch to reach inside. It heals after, but adds recovery time and potential complications.
When Is Osteotomy Needed?
Posteromedial lesions are often difficult to access with arthroscopy alone and may require a medial malleolar osteotomy (MMO). The 2024 DGOU recommendations note that MMO has a minor effect on clinical outcome compared to other factors [128].
2024 German Cartilage Registry Data
A large registry study examined MMO prevalence and consequences [129]:
- MMO improves visibility and accessibility of the talus
- But poses risk of periprocedural morbidity
- Osteotomy consolidation: 98.5% success rate
- Malreduction rate: 23.9% (lower with 3 screws vs 2 screws: 16.7% vs 32%)
MMO vs Anterior Malleolar Osteotomy (2025)
A 2025 comparison study found [130]:
- No significant difference in surgical complication rates between MMO and AMO
- Functional outcomes significantly better at 1 year with MMO
- No significant difference at 2-year follow-up
Hardware Removal
A 2025 study on adolescent athletes found 75% had osteotomy fixation screws removed due to hardware irritation [131]. Osseous union typically occurs at 6.2 weeks average.
19. Metal Resurfacing Implants (HemiCAP) - Salvage Procedure
→ When biological treatments fail, a metal cap can resurface the damaged area. Think of it like a dental crown for cartilage. It's a salvage option - a last-ditch effort before fusion/replacement.
What It Is
Metal resurfacing was proposed for large osteochondral defects not successfully treated with other surgical treatments. The HemiCAP implant is available in 15 articular component offset sizes based on medial talar dome anatomy [132].
Clinical Outcomes
77%
Resumed physical activity
4.1 mo
Mean time to return
Critical Technical Factors
Implantation accuracy is crucial [133]:
- Metal resurfacing can recover >90% of contact area when properly placed
- Warning: If implant protrudes by just 0.25 mm, peak contact stress increases by 220%
- Must be placed 0.5 mm below surrounding cartilage for proper compression
Patient-Specific Focal Resurfacing (Episealer Talus) — 2025
→ Unlike generic HemiCAP, Episealer is custom-manufactured from CT data to exactly match your talus surface. Think of it as a perfectly fitted metal patch instead of an off-the-shelf cap.
A 2025 technique paper frames Episealer as a solution for the "treatment gap": active patients with focal talar defects in an otherwise healthy joint, or patients after failed primary treatment [155].
- Indications: Focal defect in otherwise preserved joint, or failed prior biological treatment
- Contraindications: Joint-space narrowing at Van Dijk stage ≥2, defects on opposing tibial surface
A 2025 multicenter series of 15 patients at 1-year follow-up [156]:
- Significant FAOS and pain improvement
- No revision to ankle replacement at 1 year
- Promising but still very early data
Concerns
- One study reported 50% revision rate with poor prognosis in patients with increased BMI
- The 2024 DGOU guidelines state: "Metallic resurfacing of OLT can only be recommended as a second-line treatment" [128]
- The 2024 revision systematic review found older HemiCAP-type procedures had the worst complication burden: 31 failures in 111 ankles (28%) [148]
- Long-term data shows opposing cartilage does not appear to break down (12-year follow-up)
Status: Salvage procedure for failed biological treatments. Not recommended as first-line. If moving to metal, patient-specific resurfacing (Episealer) is more current than generic HemiCAP, but only if joint space and tibial side are still preserved.
20. Revision Surgery Outcomes (2024-2026)
→ "Revision" = doing surgery again after a first surgery failed. Success rates are lower than first-time surgery. The more revisions, the harder it gets.
2024 Comprehensive Revision Systematic Review (50 Studies, 806 Ankles)
The largest pooled analysis of non-primary OLT procedures to date [148]:
22%
Pooled clinical failure (revision cases)
ACI & OAT
Best revision PRO performance
28%
HemiCAP failure rate (worst)
19%
OCA failure rate in revision
- OAT remained one of the better-performing non-primary procedures
- ACI had the best pooled PRO performance but tiny dataset (only 7 ankles)
- OCA and HemiCAP looked less effective as revision procedures
BMS for Non-Primary/Revision OLT
A systematic review found concerning outcomes for revision cases [21]:
61%
Success rate (vs 82% primary)
2026 Non-Primary BMS Prospective Study
A new 2026 prospective study confirmed that non-primary BMS improves patients from baseline but still underperforms primary cases at 2 years [149].
Realignment Surgery for Failed OAT (2024)
When osteochondral autograft transplantation fails with concomitant malalignment [134]:
- Spontaneous restoration of OLT can occur after realignment surgery
- Cyst volume decreased from 0.2592 to 0.0873 cm³ (p<0.05)
- Clinical scores improved in all patients with realignment
Repeat Arthroscopy and Microfracture (2024)
A study on repeat procedures found [135]:
- Moderate satisfaction: 7.6/10
- Moderate residual pain: 4.7/10
- 21% required additional surgery after repeat microfracture
Key point: Revision success rates are consistently lower than primary surgery. The 2024 pooled review shows OAT and ACI outperform OCA and HemiCAP in revision settings, but all revision approaches carry meaningful failure risk.
21. Cryopreserved vs Fresh Osteochondral Allografts (2025)
→ Fresh allografts (from recently deceased donors) have living cells but limited shelf life. Cryopreserved (frozen) grafts last longer but cells may be dead. New tech is bridging this gap.
2025 Research Breakthrough
A 2025 study found promising results for cryopreserved grafts [136]:
- No clinically relevant biomechanical or histological differences between fresh and up to 1-year cryopreserved OCA
- Cryopreserved grafts may be a suitable alternative to fresh grafts
- Additional viability assessments still needed to validate chondrocyte survival
Traditional Understanding
Fresh allografts have been favored due to [137]:
- Increased chondrocyte viability
- Better cartilage stiffness
- Higher cellularity and matrix content at 6 months
Newer Cryopreserved Technology
- Cryopreserved Viable Osteochondral Allograft (CVOCA) can be stored frozen for up to 2 years
- Can fit to any surface contour and implanted arthroscopically
- Thin, Laser-Etched Allograft (T-LE) maintains functional components after 2 years of storage [138]
22. 3D Bioprinting & Future Technologies (2024-2025)
→ Scientists are developing ways to 3D print cartilage and bone scaffolds custom-made for each patient's defect. Still experimental but represents the future of joint repair.
2025 Breakthrough: Hybrid 3D Bioprinting
Researchers from Singapore and Manchester developed a layered scaffold that mimics natural bone and cartilage structure [139]:
- Combines soft hydrogel bioinks with hard bioceramic composite polymer
- Precisely replicates osteochondral tissue's structural and biofunctional properties
- Does not require additional growth factors
Key Technologies Under Development
- Decellularized Extracellular Matrix (dECM): Bioinks that mimic natural cartilage environment
- 4D Bioprinting: Scaffolds that change shape over time in response to stimuli
- Gene-activated Scaffolds: Deliver genetic instructions for cartilage regeneration
- Gradient Scaffolds: Transition from cartilage to bone properties in a single construct
Biphasic Scaffold Advances (2025)
PCL-based biphasic designs now offer [140]:
- Superior print precision
- Higher mechanical strength
- Sustained bioactivity without additional growth factors
- Suitable for load-bearing osteochondral regeneration
Status: Primarily laboratory research. Clinical translation ongoing. Represents promising future direction for complex OLT repair.
23. 2026 Breakthroughs: Cartilage Regeneration Drug & Emerging Therapies
→ For the first time, researchers have shown a drug can actually regenerate cartilage — not just slow its breakdown. This is potentially the biggest advance in cartilage medicine in decades.
15-PGDH Inhibitor: Cartilage Regeneration Drug (Science, March 2026)
Stanford researchers published in Science that a small-molecule inhibitor of the enzyme 15-PGDH regenerates articular cartilage in aged mice and after joint injury [173]:
- Both systemic and local injection regenerated cartilage and reduced OA-associated pain
- Mechanism: changes gene expression in existing chondrocytes — decreases hypertrophic (damaged) chondrocytes, increases matrix-synthesizing (healthy) chondrocytes
- Worked on human cartilage explants from knee replacement surgeries
- An oral 15-PGDH inhibitor is already in Phase 1 trials for age-related muscle weakness (shown safe). Cartilage-specific trial planned
→ This is the first drug shown to actually regenerate cartilage, not just slow degradation. It reprograms existing cartilage cells to build new matrix. An oral version is already proven safe in humans for another condition. A cartilage trial is next.
MACI Ankle Phase 3 Trial (MASCOT) — Now Enrolling
Vericel Corporation launched the first Phase 3 RCT comparing MACI vs bone marrow stimulation for symptomatic chondral/osteochondral lesions of the talus (ages 17-65) [174]:
- Enrollment began Q4 2025, actively recruiting in 2026
- MACI Arthro (arthroscopic delivery) already FDA-approved for knee
- Estimated >$1 billion addressable market for ankle indication
- If approved, would be the first cell-based cartilage therapy specifically indicated for the ankle
Hy2Care CartRevive Hydrogel Implant
A naturally derived dextran + hyaluronic acid injectable that gels in ~60 seconds. FDA Breakthrough Device Designation + FDA IDE approval. First US patient treatment expected early 2026 [175]:
- 46 patients treated in European clinical studies
- Currently knee-only, but represents the type of injectable hydrogel technology that could reach ankle applications
Costal Cartilage Transplantation (2025-2026)
Growing evidence for autologous costochondral transplantation for severe (Hepple V) OLT [176]:
- 27 patients with Hepple V OLT treated with rib cartilage transplantation
- Provides simultaneous cartilage AND bone in a single graft
- Avoids knee donor-site morbidity (the main OATS concern)
- Emerging as a viable new option for severe cystic OLT
CARTILAGE Journal Special Issue: "Ankle Cartilage — New Beginnings" (March 2026)
An entire special issue of CARTILAGE dedicated to ankle OLT, edited by Kerkhoffs, Kennedy, Brittberg, and Dahmen [177]. Key messages:
- The future lies in early diagnostic tools and minimally invasive interventions that halt disease before significant degeneration
- Analysis of 262 studies (11,785 patients) found no consensus on OLT classification — different terminology, non-validated systems. Standardization urgently needed [178]
- A single traumatic impact causes immediate, substantial decrease in talar cartilage mechanical properties — damage likely involves collagen fiber rupture [179]
- CT findings show limited and inconsistent correlation with perceived pain — structural damage doesn't always predict symptoms [180]
Disease-Modifying OA Drugs (DMOADs): Pipeline Update
As of 2026, no DMOADs are FDA-approved for any joint. The pipeline:
- Lorecivivint (Wnt inhibitor): Only compound reaching Phase 3; alleviates pain but no structural benefit demonstrated
- Sprifermin: Modified cartilage thickness dose-dependently, sustained at 5 years
- LNA043: Delays cartilage volume loss; Phase IIb ONWARDS trial ongoing
- 15-PGDH inhibitors: The most promising new entrant — actual cartilage regeneration, not just slowing degradation
24. Psychological Factors in OLT Outcomes (2024-2025)
→ Your mental state affects surgical outcomes. Anxiety and depression are common with chronic pain. Addressing mental health can improve physical recovery.
2024 OLT-Specific Study
A landmark 2024 study examined psychological status in OLT patients [141]:
- 48% of Hepple V OLT patients had preoperative anxiety/depression symptoms
- Both groups improved after surgery
- However, patients with preoperative psychological symptoms had poorer overall prognosis
2025 Systematic Review: Preoperative Anxiety
A meta-analysis of 115,380 orthopedic patients found [142]:
- Preoperative anxiety positively correlated with postoperative anxiety (z=0.60)
- Preoperative anxiety positively correlated with postoperative pain (z=0.22)
- Preoperative anxiety negatively correlated with joint function (z=-0.25)
- Females reported higher preoperative anxiety
Clinical Implications
- Poor preoperative mental health and depression negatively impact outcomes across orthopedic procedures
- Screening with Hospital Anxiety and Depression Scale (HADS) is recommended
- Addressing psychological factors may improve physical recovery
Takeaway: Managing pain-related anxiety and depression is an important part of comprehensive OLT treatment.
25. Platelet-Rich Fibrin (PRF) - Beyond PRP (2024-2025)
→ PRF is like PRP but contains more of the healing factors and a fibrin scaffold. Simpler preparation, no additives needed. May provide longer-lasting effects than PRP.
2024 Bulgarian Study on PRF for OLT
A retrospective study of 15 patients with stage IV OLT found [143]:
52.8→84.9
AOFAS improvement (p<0.05)
44.9→16.5
VAS pain reduction (p<0.05)
- Mean follow-up: 38.8 months
- All surgeries used same arthroscopic algorithm with PRF application
PRF + Autologous Osteochondral Transplantation (2025)
A study combining PRP with AOT in patients with OLT + chronic lateral ankle instability found [144]:
- Both groups showed significant VAS and AOFAS improvements
- AOT + PRP group showed significantly better "surface of repair tissue" scores on MOCART 2.0
PRF vs PRP
PRF provides a natural fibrin-based biomaterial scaffold rich in platelets, growth factors, and cytokines that facilitates cellular migration and proliferation essential for tissue healing.
Patient-Specific Deep Dives (April 2026)
Five focused investigations into questions directly relevant to Raymond's situation: HA optimization, revision allograft outcomes, activity guidance, bracing evidence, and clinical trial eligibility.
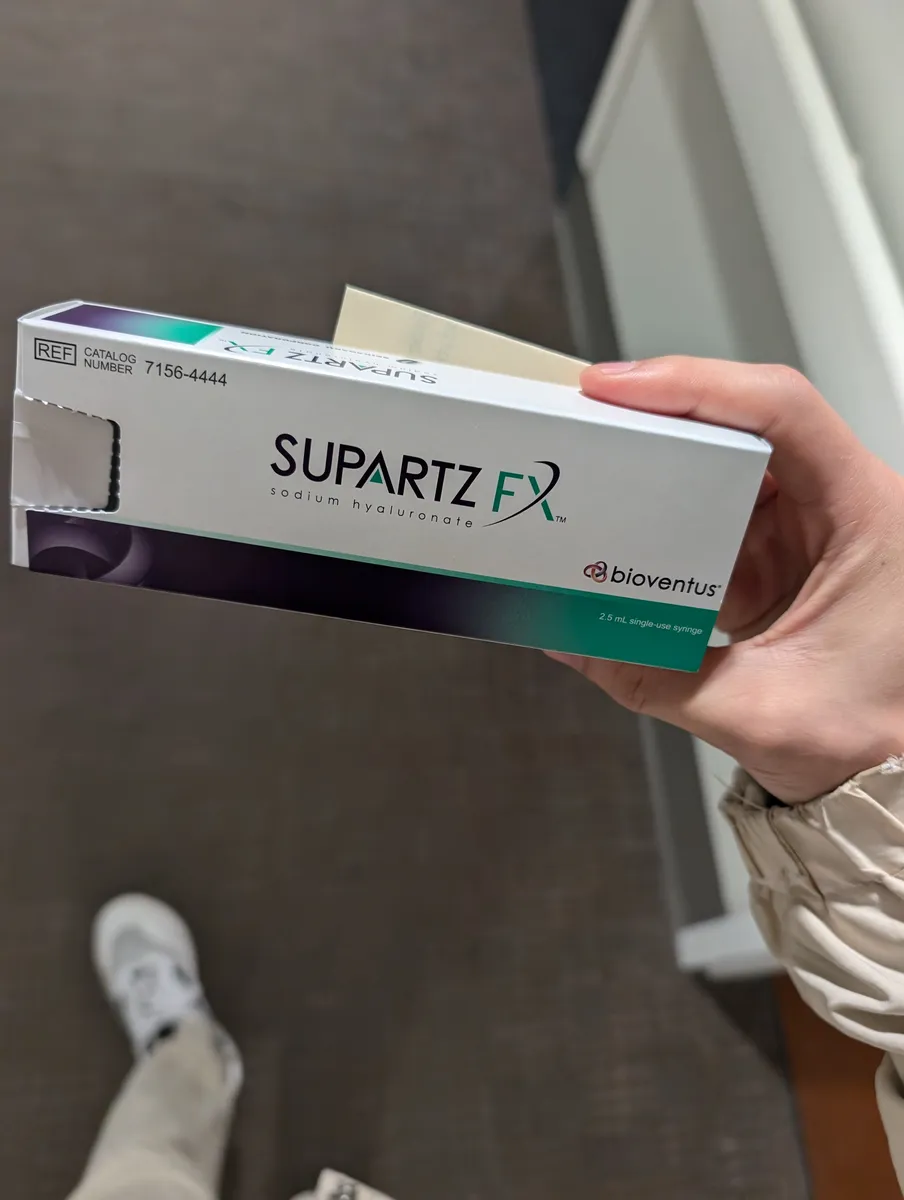
29. HA Injection Optimization (Currently on Supartz)
→ Raymond is actively on Supartz HA injections. This section covers what to expect, when to switch products, when to consider it failed, and what the data says about repeat dosing.
Expected Duration of Relief
- 4-6 months is the most consistently reported duration of meaningful pain relief per series across studies [181]
- Hwang et al. (2020): VAS significantly reduced at 12 months in HA after failed microfracture; AOFAS 50.7 → 79.9 at mean 29 months [182]
- For Hepple V cystic lesions, durability is typically at the shorter end because HA does not address subchondral bone pathology
Repeat Dosing: Efficacy Does NOT Diminish
The AMELIA Project (4 cycles of 5 HA injections each over 40 months, 306 patients) found [183]:
- Marked carry-over effect for at least 1 year after the last cycle
- Response rates to HA increased progressively throughout the study — cumulative, not diminishing
- No safety issues with repeated cycles
- Real-world data (Altman 2018, n=182,022): 95% without TKA at 5+ courses vs 71.6% for 1 course [184]
- No upper limit on number of safe series; typical interval is every 6-12 months
Product Comparison — If Supartz Wanes
Supartz is mid-MW linear HA. If response diminishes, options include:
- Cross-linked HMW products (Synvisc-One, Monovisc, Durolane): potentially longer intra-articular residence, but higher rate of post-injection flare reactions (~2.5%)
- Euflexxa: bio-fermented (non-avian), higher MW, useful if avian allergy concern
- No head-to-head ankle data shows clear product superiority — the Witteveen Cochrane review found similar modest benefits across products [185]
Predictors of Response (Han 2014)
Yonsei Medical Journal study of 40 patients found [186]:
- Positive predictors: early-stage disease (Takakura I-II), symptom duration <12 months
- NOT predictive: age, gender, fracture history, subchondral cysts
- 12-month outcomes: 40% completely satisfied, 35% dissatisfied
Failure Criteria — When to Escalate
- Set MCID thresholds before each series (AOFAS ≥10 points, VAS ≥2 points)
- If <3 months of meaningful relief per series, or ≥2 failed series → consider next-line
- Hwang 2020: 33% of HA-after-failed-microfracture patients went on to further surgery within mean 29 months
- For Hepple V: failure criteria should be tighter because HA can't fix the underlying bone problem
Off-Label Use and Insurance Reality
- FDA-approved for KNEE OA only. ALL ankle use is off-label
- Medicare LCD L39260 and most commercial insurers: HA "investigative and not covered" for ankle
- Most common outcome: denial. Patients pay cash ($300-$1200 per injection)
- Switching from Kaiser to PPO may help with appeal-based coverage but doesn't change FDA off-label status
- Boffa/Filardo 2021 systematic review (24 studies): significant benefit favoring HA vs saline at 6 months for ankle OA, GRADE evidence quality "very low" [187]
Combination Therapies Worth Considering
- HA + PRP: Mei-Dan 2012 head-to-head — PRP slightly superior to HA alone for talar OLT (VAS 4.1→0.9 PRP vs 5.6→3.1 HA) [188]
- HA + BMAC + scaffold: Buda 10-year data shows durable results for cystic OLT (this is surgical, not injection) [189]
- HA + corticosteroid: Avoid in young patient — steroid is chondrotoxic with repeated use
Long-Term Safety
- Per-injection AE rate: 1-4%; serious AEs <0.01%
- Repeated courses are at least as safe, probably safer than first course
- No cumulative toxicity, no chondrotoxicity (unlike corticosteroids)
- Main risk is opportunity cost — delaying definitive treatment of a Hepple V cystic lesion
30. Revision Allograft After Failed Primary — The Central Disagreement
→ Half Raymond's doctors recommend a second allograft (Gentile, UCSF Thuillier); half advise against it (Choung, Collman, Salk). The literature is thin but mostly supports the "advise against" camp when read rigorously.
The Only Direct Study: Gaul/Bugbee 2018
This is the single published case series of revision OCA of the ankle — from Bugbee's Scripps group, the world's largest OCA registry [190]:
84% / 65%
5-yr / 10-yr revision graft survivorship
50%
Required further surgery
6.7 yr
Mean time to failure
- 20 patients (the entire ankle revision OCA literature)
- 57% reported moderate-to-severe pain even among surviving grafts
- Of failures: 4 went to arthrodesis, 1 to TAR, 1 to amputation
- Comparator: primary talar OCA shows 89% 10-year survival — revision is roughly 24 percentage points worse
Why Primary Allografts Fail (Williams Histopathology Study)
8 retrieved failed talar allografts examined [191]:
- Mean time to failure: 31 months (most failures are early, not late)
- All grafts: subchondral collapse and lucencies
- Histology: substantial loss of glycosaminoglycans, osteocalcin "nearly absent" (non-viable bone)
- CD4+ and CD8+ T-cell infiltrates at the graft-host interface — failure is primarily biologic (immune-mediated), not mechanical
→ Raymond's 12-year pain-free interval is unusually long — most failures happen in the first 3-5 years. This suggests his biology supported initial incorporation, which is favorable but doesn't tell us how a second graft will fare.
Head-to-Head: Revision Options Comparison
Shah systematic review of failed bulk talar allograft revision [192]:
77.3%
Ankle arthrodesis satisfactory
50%
Revision allograft satisfactory
Fusion outperforms revision allograft by ~27 percentage points in this systematic review.
The "Bulk Dead Bone" Problem (Van Dijk Editorial)
Van Dijk's commentary [193] argues:
- Most of the failed graft remains dead bone (per Williams histopathology)
- Bulk grafts compromise future salvage to fusion or TAR
- Each prior procedure adds ~2.5x failure risk per multivariable analysis
- Revision typically requires a LARGER graft (debridement back to bleeding bone)
- Larger grafts (>8 cm²): 2.12x failure risk (Bugbee 2025, n=560 knees) [194]
Salvage After Failed Revision OCA (Gaul 2019)
What happens if revision allograft fails [195]:
- 13 fusion patients: 88% satisfied, AAOS-FAM 83, but 63% reported continued ankle problems
- 11 TAR patients: only 50% satisfied, 40% continued problems
- Both fusion and TAR after failed OCA perform worse than primary fusion/TAR — the joint is compromised regardless of salvage strategy
Immunologic Considerations
- OCA is NOT HLA or ABO matched in clinical practice; chondrocytes are immunoprivileged [196]
- Blood type mismatch study (n=103): no association between mismatch and failure
- No published data on whether prior allograft exposure sensitizes for a second allograft
- The 12-year pain-free interval argues against aggressive immune response in this specific patient
Most Defensible Framing for Decision-Making
"Revision allograft is the only joint-preserving option but carries roughly 35% chance of outright failure at 10 years and only ~50% chance of being satisfied with the result even if the graft survives. Fusion outperforms it in direct comparison (77% vs 50% satisfactory). The central trade-off: 'one more shot at preserving motion with high uncertainty' vs 'committing to a definitive but non-motion-preserving salvage now with better-documented outcomes.'"
31. Activity-Specific Evidence for OLT
→ What activities are actually safe vs harmful with this lesion? The answer comes from biomechanics (joint loading) + return-to-sport data after surgery + cartilage response to mechanical loading.
Talar Joint Loading Hierarchy (Multiples of Body Weight)
~0
Swimming, cycling (seated), kayak
5.6-6.1x
Walking joint reaction force
[197]
~11x
Running joint force (+94% vs walking)
[198]
11.5x
Heavy squat (90% 1RM)
[199]
Defect Biomechanics: Where Raymond Sits (FEA Studies)
From Ruan et al. 2023 finite element analysis [200]:
- Defect area >1.0-1.5 cm² → peak stress migrates to lesion edges
- Defect depth ≥3 mm (into subchondral bone) → significant stress increase, continues rising up to 10 mm
- 2 cm² defect: peak stress doubles vs 0.5 cm²
- Stress hierarchy by motion: dorsiflexion > internal rotation > inversion > external rotation > plantarflexion > eversion
Raymond's 1.5 cm² cartilage + 10 mm cyst exceeds every biomechanical threshold identified as destabilizing. Each impact cycle loads a zone pre-disposed to edge progression.
Return to Sport After Talar OCA (Fiske/Bugbee 2024)
Most directly relevant to Raymond — 36 ankles, mean 9.2-year follow-up [201]:
- 66.7% participated in sports/recreation post-op; 50% still participating at latest follow-up
- Competitive athletes: 73.9% returned at some point, 65.2% still participating
- Graft survivorship: 94.3% at 5 years, 85.3% at 10 years
- 79.4% satisfied or extremely satisfied
- Pattern: post-OCA patients shift away from high-impact and contact sports toward low-impact recreation
Cartilage Response to Loading: "Use It Appropriately"
- Moderate cyclic loading is anabolic — upregulates collagen II, suppresses MMPs (TRPV4/CITED2 pathways) [202]
- Disuse causes cartilage atrophy in humans within 7 weeks of reduced weight-bearing
- Disuse atrophy is reversible but accelerates post-traumatic OA progression if maintained
- End-stage ankle OA cohorts who stayed active chose predominantly cycling and swimming [203]
Activity Recommendations Summary
- Green light: Swimming, stationary cycling, elliptical, rowing, flat-water kayak/SUP, Pilates, gentle yoga, leg press, RDL, hip thrust, calf raises, walking on level ground
- Yellow light: Hiking with trekking poles + light pack on stable trails, outdoor cycling, moderate strength training, surfing small waves, low-amplitude plyometrics
- Red light: Running (road or trail), basketball, soccer/tennis/pickleball (cutting sports load inversion/rotation = #2 and #3 highest stress motions), jumping sports, heavy 1RM squats in max dorsiflexion
Footwear: The "Running Shoes Indoors" Recommendation
- Indoor surfaces (hardwood, tile, concrete) have effectively zero compliance — the shoe is the entire cushioning system
- Best choice: moderate stack (25-32 mm), moderate drop (6-10 mm), stable heel counter
- Maximalist cushioning (Hoka, >35 mm stack): can paradoxically increase leg stiffness and impact loading [204]
- Rocker geometry reduces peak ankle dorsiflexion demands — favorable since dorsiflexion is the highest-stress talar position
32. Ankle Bracing & Orthotics for OLT
→ Dr. Salk recommended bracing. The honest answer: there is no published RCT, systematic review, or clinical guideline specifically testing bracing for OLT. The recommendation is biomechanically plausible but not evidence-based in the strict sense.
Direct Evidence for Bracing in OLT: Almost None
- No published RCT, systematic review, or guideline on bracing for OLT specifically
- Van Diepen systematic review of nonoperative OLT management: only 8 articles even mentioned bracing/strapping; none isolated bracing as an independent variable [205]
- Authors state: "no radiological results can demonstrate improvement of the lesion due to immobilization"
- Bracing is recommended as symptomatic and protective, not disease-modifying
Indirect Evidence: Biomechanical Mechanisms
- Lace-up brace reduces tibiotalar ROM (42.7° → 38.7°) and AP translation (16.9 mm → 15.0 mm), reducing shear across talar dome [206]
- Semi-rigid brace 3D stress-MRI study: increased cartilage contact area in plantarflexion/supination — force spread over more cartilage, reducing focal peak pressures [207]
- No conventional ankle brace selectively offloads medial vs lateral talar dome — that requires a wedge orthotic
Best-Evidenced Device for Late-Stage Tibiotalar Pathology
Corr et al. 2022 prospective cohort — Arizona Brace (gauntlet AFO), 102 patients [208]:
- Pain with walking dropped from 63.6 → 44.6 on VAS (p=0.003) — clinically meaningful
- 62.7% had discontinued use at follow-up — compliance is the central limitation
- This is the best available prospective evidence for any brace in degenerative tibiotalar disease
Custom Orthotics: Foot Type Matters
- Cavovarus foot (high arch, drives medial talar overload): lateral heel/forefoot wedge or full-length orthotic with lateral posting
- Planovalgus foot: medial heel wedge or UCBL orthosis
- Off-the-shelf wedges without biomechanical assessment are a gamble — podiatric exam needed
- This is direct from Manoli's "subtle cavus" protocol — lateral wedging is standard of care for cavus-driven medial talar pathology
Side Effects: NOT a Concern
- Long-term brace wear does NOT impair peroneal latency during sudden inversion
- Proprioception is not impaired by ankle bracing in CAI meta-analyses [209]
- Muscles are if anything more active under a brace during gait — counters atrophy hypothesis
- APTA Clinical Practice Guideline 2021: bracing for 6-12 months combined with exercise is recommended for chronic ankle instability
Practical Recommendations
- First-line: Lace-up (ASO) or semi-rigid stirrup (Aircast A60) — best evidence for sprain prevention; minimal compliance issues
- For pain reduction: Bauerfeind MalleoTrain compression sleeve (gentlest, pain-focused)
- If pain progresses or first-line fails: Escalate to Arizona Brace (gauntlet AFO) — best evidence for late-stage tibiotalar pathology
- Rocker-sole shoe is the single footwear feature with the best evidence for ankle arthritis
- Pair with PT — bracing alone is inferior to bracing + structured exercise in every trial
- Custom orthotic with appropriate posting based on foot type assessment
33. Clinical Trial Eligibility (April 2026)
→ The active US OLT/ankle cartilage trial landscape is very narrow. Only one trial directly targets joint-preserving treatment for OLT — and the closest site is in Texas, not California.
MASCOT (NCT06915233) — The Top Priority Trial
Vericel's Phase 3 RCT of MACI vs bone marrow stimulation for symptomatic chondral/osteochondral defects of the ankle [210]:
- Sponsor: Vericel Corporation
- Phase: Phase 3, randomized 2:1, n=309 target
- Status: Recruiting (verified March 2026)
- Primary endpoint: FAOS Pain + Function at Week 104
- Eligibility: Age 17-65, ICRS grade 3-4 talar lesion, ≥1.2 cm²
- Key exclusion: Bony defect depth >5 mm. Raymond's 10 mm cyst likely disqualifies him — this is the critical question.
- Sites (none in California): Washington DC, Wichita KS, Houston TX, San Antonio TX
- Action: Call Vericel Clinical Affairs at 978-347-2876 or clinicalhotline@vcel.com to confirm eligibility re: cyst depth and prior allograft
Closest Bay Area Site (Any Talus Trial)
Paragon 28 Patient Specific Talus Spacer Post-Approval Study (NCT05364606) [211]:
- Site: Redwood Orthopaedics, Santa Rosa, CA — PI Dr. Thomas Chang, DPM, 707-544-3400
- Indication: AVN of talus (NOT OLT) — Raymond likely doesn't qualify
- BUT: Dr. Chang is a recognized foot/ankle researcher; worth a call even if trial isn't a fit — he may know of other ankle cartilage trials or off-label options in the Bay Area
Total Talus Replacement Trials (Joint-Sacrificing)
- Restor3d PROCLAIM (NCT06311331): Post-approval study, OrthoCarolina Charlotte NC. Enrolling by invitation. AVN/large talar OCD indication [212]
- 3DTalar Registry (NCT03965143): University of Missouri. AVN only
- Paragon 28 SMART Total Talus IDE: FDA-approved Aug 2023 but no CT.gov registration as of April 2026
- All are joint-sacrificing — contradicts Raymond's stated preference for joint preservation
Future Trials to Watch
- 15-PGDH inhibitor (MF-300, Epirium Bio): Phase 1 in healthy volunteers complete (sarcopenia indication). No OA/cartilage trial registered. Earliest cartilage trial: 2027-2028
- Hy2Care CartRevive: FDA IDE approved April 2025. First US patient expected early 2026. Knee only, no ankle expansion announced
- Stanford 15-PGDH lab (Helen Blau): Direct contact may yield information about future cartilage trial plans
Bay Area Academic Centers: Trial Status
- UCSF: No active OLT/ankle cartilage trial. Worth contacting Foot & Ankle service for off-trial expert consultation
- Stanford: Drs. Loretta Chou, Geoffrey Abrams publish on OLT but no active trial. Worth direct contact for unregistered studies or compassionate access
- UC Davis, Kaiser, Cedars-Sinai, Scripps, Hoag: No enrolling OLT trials identified
International Options (If Highly Motivated)
- NCT03347877: Autologous osteo-periosteal cylinder graft specifically for Hepple V OLT — Peking University Third Hospital, Beijing. Enrolling by invitation
- Amsterdam UMC (Dahmen / Kerkhoffs): World leaders in OLT research, deepest trial pipeline
- Ospedale Galeazzi, Italy: AMIC for talus trial (NCT03371121)
Recommended Next Steps
- Call Vericel at 978-347-2876 to clarify MASCOT eligibility (cyst depth, prior allograft)
- If MASCOT doesn't fit, contact Stanford or UCSF Foot & Ankle for off-trial expert consultation
- Call Dr. Thomas Chang at Redwood Orthopaedics (707-544-3400) for Bay Area trial knowledge
- Set up CT.gov email alerts for "osteochondral talus," "MACI ankle," "ankle cartilage," "talus cartilage"
- Contact Epirium Bio for MF-300 cartilage trial timing (long shot but worth a note)